
Targeting galectin-9 to unlock new possibilities in cancer treatment
Gallop Oncology is a clinical-stage biopharmaceutical company committed to transforming treatment paradigms for myeloid malignancies
We are advancing a novel approach where efficacy, safety, and durability converge. Our lead candidate, LYT-200, is the most advanced candidate targeting galectin-9, an important oncogenic driver and potent immunosuppressor in cancer, offering a differentiated strategy to address some of the most challenging cancers.
Gallop Oncology was founded by and is currently a wholly-owned subsidiary of PureTech Health.
Scientific Advisory Board
-

Steven Rosen, MD
Executive Vice President and Director emeritus, City of Hope Beckman Research Institute and Cancer Center
-

Eytan Stein, MD
Chief, Leukemia Service and Director, Program for Drug Development in Leukemia, Division of Hematologic Malignancies of Memorial Sloan Kettering Cancer Center
-

Joseph Jurcic, MD
Director, Hematologic Malignancies Section of the Division of Hematology/ Oncology, Colombia University
Galectin-9:
A Groundbreaking Target
Galectin-9 is an important oncogenic driver and potent immunosuppressor in cancer, offering a differentiated strategy to address some of the most challenging cancers. It promotes multiple immunosuppressive pathways, and blocking galectin-9 results in tumor cell death as well as induction of anti-tumor immunity in the context of myeloid malignancies.
LYT-200:
A Mutation-Agnostic, Dual Mechanism of Action
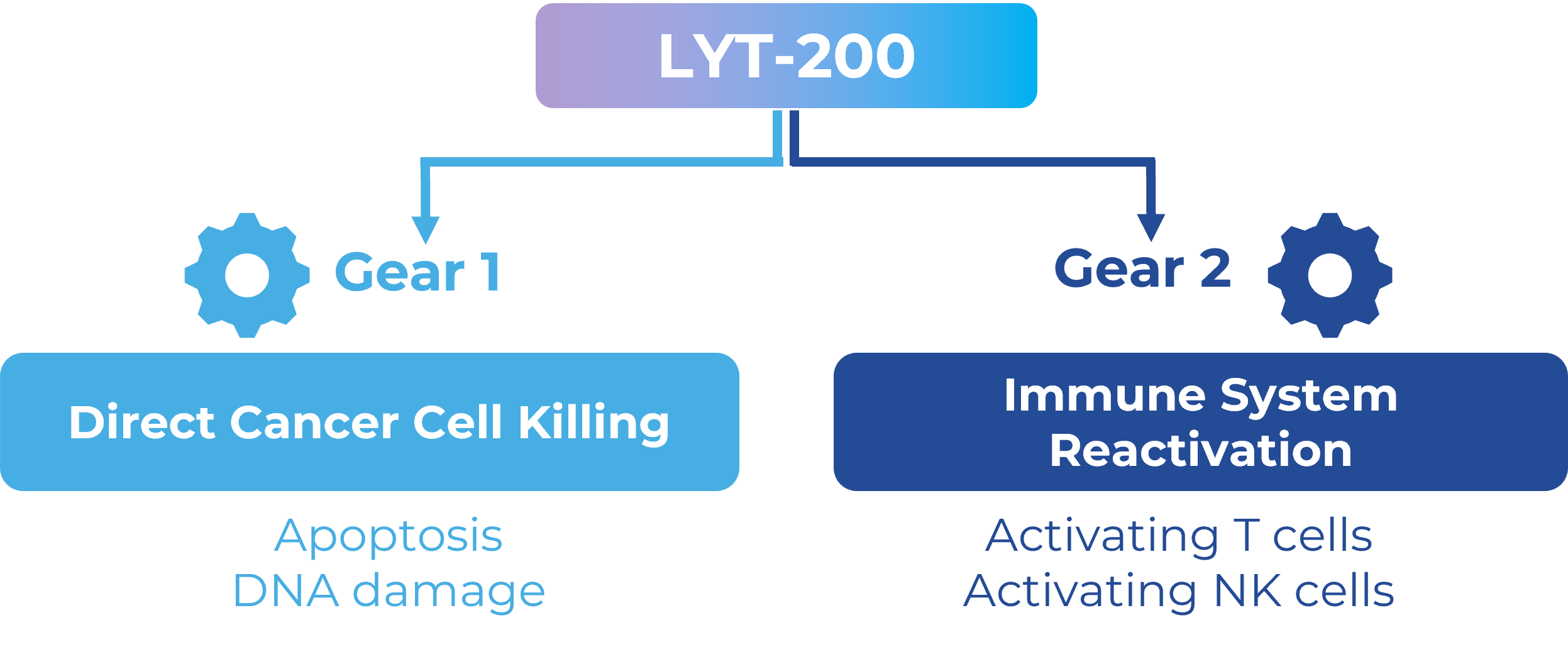
LYT-200 is a fully human IgG4 monoclonal antibody and, to our knowledge, the most advanced clinical program targeting galectin-9. Targeting galectin-9 offers a dual mechanism of action:
Direct Cancer Cell Killing: Inhibiting galectin-9 induces DNA damage and apoptosis in cancer cells.
Immune System Reactivation: Galectin-9 disrupts immune surveillance by binding to multiple receptor binding partners, including CD45, CD44, 4-1BB, PD-1, DR3, VISTA, CD206, Dectin-1 etc. , facilitating immunosuppression. Inhibition of galectin-9 removes this barrier, restoring immune system efficacy against tumors. This mutation-agnostic approach supports potential use of LYT-200 as both a monotherapy and in combination with other anti-cancer therapies, depending on the cancer type, treatment setting, and line of treatment.
Transforming Oncology Treatment
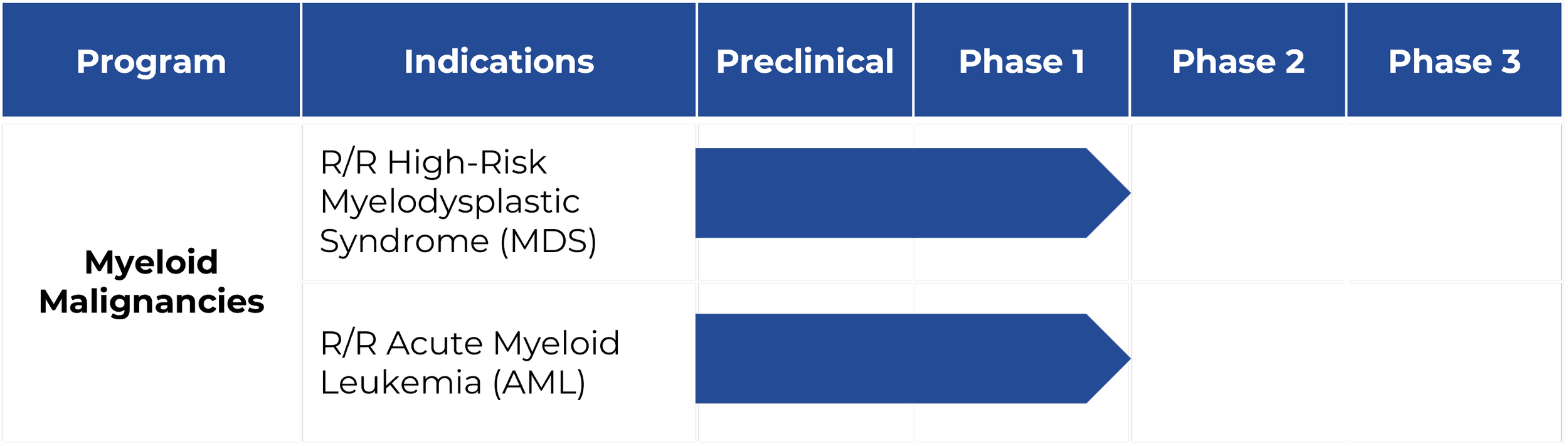
LYT-200 has been evaluated in a Phase 1b trial as a monotherapy and in combination regimens in two heavily pretreated patient populations:
Relapsed/refractory high-risk myelodysplastic syndrome: LYT-200 in combination with a hypomethylating agent (azacitidine or decitabine)
Relapsed/refractory acute myleoid leukemia: LYT-200 in combination with venetoclax and a hypomethylating agent (azacitidine or decitabine)
LYT-200 has been granted Fast Track designation and Orphan Drug designation from the FDA for the treatment of acute myeloid leukemia.
Addressing Unmet Clinical Needs
-
Myelodysplastic syndromes (MDS) are a group of serious blood cancers characterized by ineffective blood cell production in the bone marrow, leading to anemia, infections, and bleeding complications. [1,2] MDS affects approximately 60,000–170,000 people in the United States, with an estimated 30–40% of patients diagnosed with the more aggressive form of the disease known as high-risk (HR) MDS.[1,3] HR-MDS is associated with poor outcomes, with median survival typically less than two years following diagnosis, and approximately 30% of patients progressing to acute myeloid leukemia (AML). [2-4]
The current standard frontline treatment for HR-MDS are hypomethylating agents (HMAs), such as azacitidine and decitabine; however, most patients do not respond to these therapies or eventually stop benefiting from them.7 Once the disease becomes relapsed or refractory (R/R), outcomes are especially poor, with survival often limited to only a few months.[5,6]
Treatment options for patients with R/R HR-MDS remain very limited. Only one therapy has been approved specifically for this setting in the past two decades, and it targets only a small subset of patients (~3–5%) with a specific genetic mutation.[5] As a result, there remains a significant need for new treatment approaches for patients with HR-MDS.
American Cancer Society. (2023). What Is Myelodysplastic Syndrome? Retrieved from https://www.cancer.org
National Comprehensive Cancer Network. (2024). NCCN Clinical Practice Guidelines in Oncology: Myelodysplastic Syndromes (Version 2.2024). Retrieved from https://www.nccn.org
Greenberg, P. L., Tuechler, H., Schanz, J., Sanz, G., Garcia-Manero, G., Solé, F., Bennett, J. M., Bowen, D., Fenaux, P., Dreyfus, F., Kantarjian, H., Kuendgen, A., Levis, A., Malcovati, L., Cazzola, M., & Haase, D. (2012). Revised International Prognostic Scoring System for myelodysplastic syndromes. Blood, 120(12), 2454–2465. https://doi.org/10.1182/blood-2012-03-420489
Ma, X. (2012). Epidemiology of myelodysplastic syndromes. The American Journal of Medicine, 125(7 Suppl), S2–S5. https://doi.org/10.1016/j.amjmed.2012.04.014
Garcia-Manero, G., Fenaux, P., Al-Kali, A., Baer, M. R., Sekeres, M. A., Roboz, G. J., et al. (2016). Rigosertib versus best supportive care for patients with high-risk myelodysplastic syndromes after failure of hypomethylating drugs (ONTIME): A randomised, controlled, phase 3 trial. Lancet Oncology, 17(4), 496–508. https://doi.org/10.1016/S1470-2045(16)00009-7
Prébet, T., Gore, S. D., Esterni, B., Gardin, C., Itzykson, R., Thepot, S., Quesnel, B., Dreyfus, F., Beyne-Rauzy, O., Vey, N., Recher, C., Adès, L., Fenaux, P., & Groupe Francophone des Myélodysplasies. (2011). Outcome of patients with higher-risk myelodysplastic syndromes after azacitidine treatment failure. Journal of Clinical Oncology, 29(24), 3322–3327. https://doi.org/10.1200/JCO.2011.35.8135
-
Acute myeloid leukemia (AML) is an aggressive blood cancer characterized by the rapid growth of abnormal myeloid blast cells in the bone marrow and blood. It is the most common form of acute leukemia in adults, with a five-year survival rate of less than 30%.[1] Despite available therapies, many patients relapse or fail to respond, and outcomes are especially poor in the relapsed/refractory setting. Around 450,000 people globally are living with AML.[1]
AML is an area of urgent medical need where new therapies with improved safety, efficacy, and durability or responses are critical. Importantly, the incidence of AML is increasing, and the market is expected to grow to $6 billion annually by 2030,[2] underscoring the scale of the opportunity to bring forward therapies that are not only more effective but also applicable across a broader segment of patients.
Acute Myeloid Leukemia - Cancer Stat Facts. (n.d.). National Cancer Institute
Grand View Research, Acute Myeloid Leukemia Treatment Market Size, Share & Trends Analysis Report By Disease, By Treatment (Chemotherapy, Targeted Therapy, Immunotherapy), By Route of Administration, By End Use, By Region, And Segment Forecasts, 2025-2030
